DDK Scientific
Products
Clean Rooms
Isolators
UV Light Disinfection
Stem Cell Laboratories
Clean Rooms
A room in which the concentration of airborne particles is controlled to meet a specified airborne particulate cleanliness class. Microorganisms in the environment are monitored so that a microbial level for air, surface, and personnel gear are not exceeded for a specified cleanliness class.
Custom designed portable Clean Rooms to meet any need.
Other Applications
of Clean Room Technology
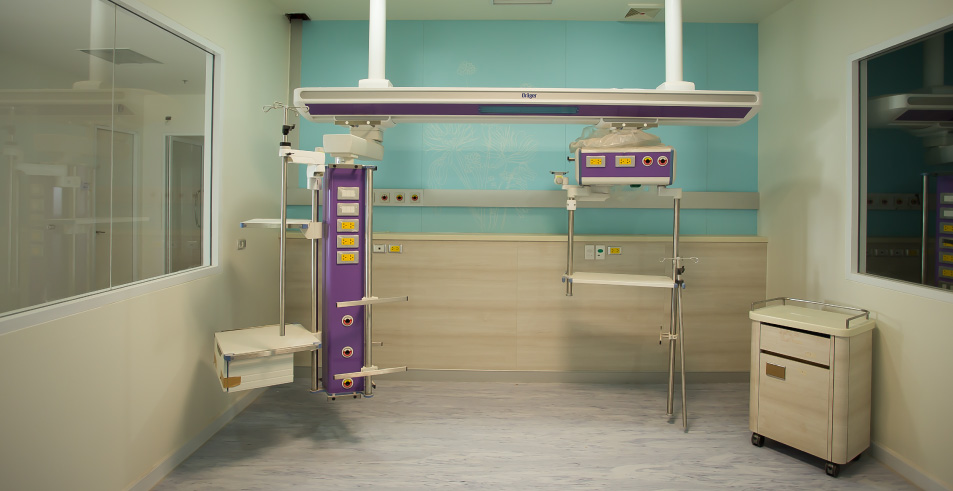
Pharmaceutical Applications
- Clean Rooms
- Portable Rooms
- Custom Isolators
- USP <797> Compliant Isolators
Research laboratories
- Design
- Construction Supervision
- Validation
- SOPs
- Protocols
Isolators
Explosion proof isolator under negative pressure
USP <797> Compliant Isolators
Compounding Aseptic Isolator (CAI)
A form of isolator specifically designed for compounding pharmaceutical ingredients or preparations. It is designed to maintain an aseptic compounding environment within the isolator through the compounding and material transfer processes. Air exchange into the isolator from the surrounding environment should not occur unless the air first passes through a microbial retentive filter (HEPA minimum)
Compounding Aseptic Containment Isolator (CACI)
A compounding Aseptic Containment Isolator designed to provide worker protection from exposure to undesirable levels of airborne drug throughout the compounding and material transfer processes and to provide an aseptic environment for compounding sterile preparations. Air exchange with the surrounding environment should not occur unless the air is first passed through a microbial retentive filter (HEPA minimum) system capable of containing airborne concentrations of the physical size and state of the drug being compounded. Where volatile hazardous drugs are prepared, the exhaust air from the isolator should be appropriately removed by properly designed building ventilation.
UV Light Disinfection
A Clean Technology
Ultra Violet (UV) light, energy found in a part of the electromagnetic spectrum of sun light with wave lengths of 200 – 300 nm is known for its germicidal effects (lethal to microorganisms). By disrupting the reproductive mechanism (DNA) of bacteria, viruses, yeast or mold spores, the organisms are inactivated, thereby eliminating them as agents of disease, spoilage and unwanted biological growth.
Custom Designed Equipment
In house, we design and fabricate top quality UV light equipment to exactly meet your needs for surface, air and liquids disinfections, with NIST traceable validation and documentation.

Unique Features UV Sterilization Offers
- No disinfections by-products
- No EPA reporting
- Non-corrosive
- Low maintenance
Unique Benefits UV Sterilization Offers
- Fastest
- Consistent
- Efficient
- Cost effective
Validation Documentation
UTILIZATION OF NIST TRACEABLE VALIDATION INSTRUMENTATION
Included in the Validation are the Following Features
- Validation of design criteria
- Installation qualifications
- Operating qualifications
- Performance qualifications
- Validation of accurate UV dose
- Detailed documentation of measurements
Pharmaceutical Applications
Environmental
- Air Disinfection
- Water Disinfection
- Dechlorination
Surface Disinfection
- Packages
- Containers
- Screw Caps
- Aseptic Packaging
- Others
Liquid Sterilizing
- Blood Plasma
- Ultra Pure Water
- Water for Injection
- Others
Food Processing Applications
Environmental
- Air Disinfection
- Water Disinfection
- Dechlorination
Surface Disinfection
- Packages
- Containers
- Screw Caps
- Aseptic Packaging
- Bread / Bakery Products
- Fruits & Vegetables
Liquid Sterilizing
- Process Water
- Fruit Juice
- Others
Stem Cell Research Laboratories
Good Manufacturing Practice (GMP)
Turn-Key Projects
Stem cell research offers the potential to dramatically expand the understanding of regenerative cellular functions and processes, and therefore allows the development of techniques for tissue repair and regeneration. Clinical applications of stem cells promise to vastly improve the ability to successfully address life-threatening diseases, such as heart and vascular disease, diabetes, Parkinson’s disease, Alzheimer’s disease and stroke, and also debilitating physical injuries, such as spinal cord injuries, peripheral nerve injuries and burns.
What are Stem Cells?
They are unspecialized cells that, when they divide, can self-renew, but at the same time can also become cells with special functions. For instance, mature blood cells of all lineages (red and white), cartilage, bone, muscle, beating cells of the heart muscle, liver cells or insulin-producing cells of the pancreas.
Good Manufacturing Practice (GMP) is a national standard for the production of pharmaceuticals, prescribed by the United States Food and Drug Administration (FDA) that assures safe and effective drugs.
The roots for this standard date back more than 100 years. To assure that the latest standard is applied, the term current Good Manufacturing Practice (cGMP) is used. Stem cell applications are cellular drug applications, and are regulated by very similar standards as pharmaceuticals.
Safe and effective cellular drug manufacturing requires control and standardization of a large range of parameters in the manufacturing process. For such purposes, Standard Operating Procedures (SOPs) must be implemented, covering all aspects of the production / manufacturing process. Quality Control (QC) and Quality Assurance (QA) units, completely separate from each other have to be established to make sure that the final product meets quality requirements. Manufacturing should be unidirectional and transparent, all reagents and products have to be traceable, personnel must be trained and tested for proficiency, and finally, a GMP facility, a controlled environment for product manufacturing, has to be used.
A GMP facility is a controlled environment that allows the manufacturing of a safe and potent pharmaceutical ready for administration into humans.
This controlled environment is described, in its basic layout, in the Code of Federal Regulations, 21CFR Part 210, and 211. Prerequisite is a Class 10,000 (10,000 particles/cubic foot) clean room with an ante-room. The room should be positively pressurized toward the outside environment. Some flexibility is built into the GMP regulations; therefore, good judgment in GMP facility design is required. This incorporates a layout that keeps in mind gowning and de-gowning of personnel, product, personnel and waste flow, change-over from one product to another, environmental cleaning and monitoring, equipment and facility monitoring and finally redundancy to continue manufacturing in case of equipment or HVAC failure.



